Neuroyer's Anna FDA Approves World's First VR-Based Diagnostic Medical Device for Dizziness
Aug 29, 2025
|
Dizziness is a disease in which the number of patients continues to increase with the entry of an ultra-aging society. Although it is a common disease that causes great inconvenience in daily life, existing diagnostic equipment is only used in some medical institutions due to high entry barriers such as expensive device costs and securing dedicated space. As a result, there was a limitation in access to medical care that many patients with dizziness did not receive a precise diagnosis at the appropriate time.
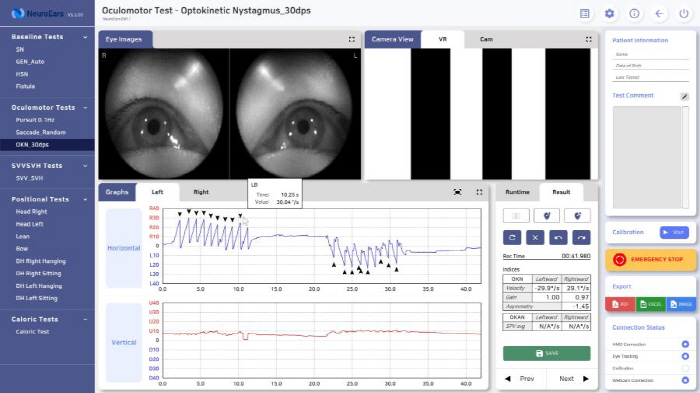
To solve this problem, Neuroaids Co., Ltd. has developed 'Neuroaes Anna', which can diagnose dizziness using VR devices. By utilizing VR devices that have already been commercialized in the market, deployment costs can be reduced by up to 80% or more compared to existing equipment. In addition, tests can be performed in general clinics without large-scale equipment, making it easy to introduce private hospitals without cost and space constraints.
|
'Neurores Anna' has been introduced to more than 120 medical institutions in Korea and has been proven to have excellent clinical effects, and is widely used from major university hospitals in Korea to private clinics. It has laid the foundation for entering the global market by completing successful Proof of Concept (PoC) overseas such as Vietnam and Qatar.
With the FDA approval, Neuroaids Co., Ltd. plans to enter the U.S., the world's largest medical device market. In addition, it plans to complete an integrated dizziness solution that encompasses measurement, diagnosis, and rehabilitation by adding an AI-based diagnostic assistance system and a customized rehabilitation program in the future.
Hong Seong-kwang, Chief Technology Officer (Professor of Otolaryngology at Hallym University Sacred Heart Hospital), said "This FDA approval goes beyond simple product certification and is globally recognized for innovation in the diagnostic paradigm for dizziness using VR technology. Based on this, we will strive to develop further upgraded follow-up product technology."
CEO Seo Kyu-won expressed his ambition, saying, "In the future, we will contribute to improving the diagnostic efficiency of medical staff and improving the quality of life of patients with patient-centered innovative technology, and finally achieve the commercialization of digital treatment for dizziness." He then stressed that "New Royals will grow into a leading company in global healthcare innovation based on this achievement.".
Meanwhile, Neuroaids Co., Ltd. is a subsidiary of Hallym University Technology Holding Company established in 2021 and specializes in VR-based medical devices. Founded under the leadership of Professor Seo Kyu-won of Hallym University's Industry-Academic Cooperation Group and Professor Hong Sung-kwang of Hallym University Sacred Heart Hospital, it focuses on developing innovative technologies to solve fundamental problems in the medical field for diagnosing dizziness.
|
This article was translated by Naver AI translator.