IBS Researchers Find Out How mRNA Vaccines Work
Apr 04, 2025
The working principle of the ribonucleic acid (mRNA) vaccine, which played a role of 'relief pitcher' during the COVID-19 pandemic, has been revealed by a domestic research team.
A research team led by Kim Bit-na-ri (chair professor of life sciences at Seoul National University) of the RNA research team at the Institute of Basic Science (IBS) found a protein group that controls the intracellular delivery and degradation of mRNA vaccines and identified their operation for the first time. The results of the study were published in the world's most prestigious journal Science on the 4th.
mRNA-based technology is well known as a COVID-19 vaccine, and it is attracting attention because it can be used in various ways, such as cancer vaccines, immunization, and gene therapy, as well as in response to infectious diseases. In particular, it has grown into an innovative treatment platform through mRNA synthesis techniques and the development of lipid nanoparticles, an in vivo delivery material.
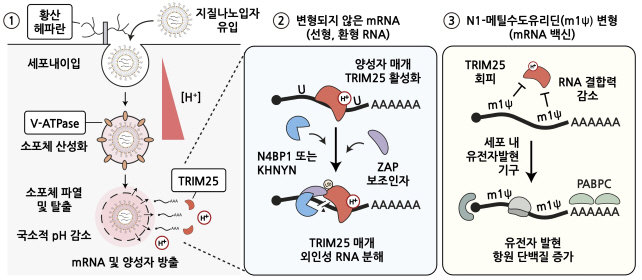
However, it is not sufficiently known how therapeutic RNA is operated and regulated in the body. In addition, it was not clear what the principle of the N1-methylsudoruridine modified base, which is the main driver of the COVID-19 vaccine, was.
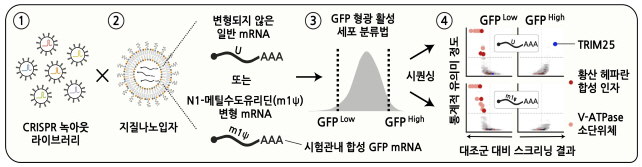
In this regard, the IBS research team closely conducted 'knockout screening' using gene scissors to find intracellular factors that control mRNA. CRISPR knockout screening is a technology that uses gene scissors to individually knock out (remove) genes and then analyze genes that affect specific traits or cell responses. In this study, a CRISPR library containing about 20,000 genes was used to screen cell factors regulating mRNA vaccines at the genome level.
Through this, the research team identified key protein factors and regulatory pathways necessary for mRNA to be delivered and introduced into cells. We also found for the first time an important role of proton ions in alerting invasion of external RNA, along with major inhibitory factors for RNA therapeutic agents. In addition, it was discovered that the TRIM25 protein, which binds and removes mRNA, significantly reduces its binding power to the N1-methylcouridine-modified base, preventing mRNA from being cleaved and degraded. We came to understand the factors and principles that could improve the efficacy and stability of the COVID-19 mRNA vaccine.
Director of IBS Noh Do-young said "This study is significant in that it has laid the theoretical foundation for enhancing the efficacy and stability of mRNA treatments by revealing the principle of intracellular operation of mRNA vaccines for the first time."
Director Kim Bit-nari said, `It is expected to provide new research directions not only for RNA but also for immunity and cell signaling by further expanding our understanding of the cell's defense mechanisms against external invaders.'
A research team led by Kim Bit-na-ri (chair professor of life sciences at Seoul National University) of the RNA research team at the Institute of Basic Science (IBS) found a protein group that controls the intracellular delivery and degradation of mRNA vaccines and identified their operation for the first time. The results of the study were published in the world's most prestigious journal Science on the 4th.
mRNA-based technology is well known as a COVID-19 vaccine, and it is attracting attention because it can be used in various ways, such as cancer vaccines, immunization, and gene therapy, as well as in response to infectious diseases. In particular, it has grown into an innovative treatment platform through mRNA synthesis techniques and the development of lipid nanoparticles, an in vivo delivery material.
However, it is not sufficiently known how therapeutic RNA is operated and regulated in the body. In addition, it was not clear what the principle of the N1-methylsudoruridine modified base, which is the main driver of the COVID-19 vaccine, was.
|
|
Director of IBS Noh Do-young said "This study is significant in that it has laid the theoretical foundation for enhancing the efficacy and stability of mRNA treatments by revealing the principle of intracellular operation of mRNA vaccines for the first time."
Director Kim Bit-nari said, `It is expected to provide new research directions not only for RNA but also for immunity and cell signaling by further expanding our understanding of the cell's defense mechanisms against external invaders.'
This article was translated by Naver AI translator.